WOUND BED
Assessment of the wound bed includes observing and recording the tissue types, levels of exudate and the presence or absence of local and/or systemic wound infection. A wound will consist of different tissue types at different stages of healing. These tissue types are often described by colour – Black, Yellow, Red and Pink and tools such as the “Wound Healing Continuum” (Gray et al, 2010) have been developed. This tool incorporates intermediate colour combinations between the four key colours (see Figure 3.8). This practical approach to everyday wound care enables the practitioner to approach wound assessment logically and systematically.
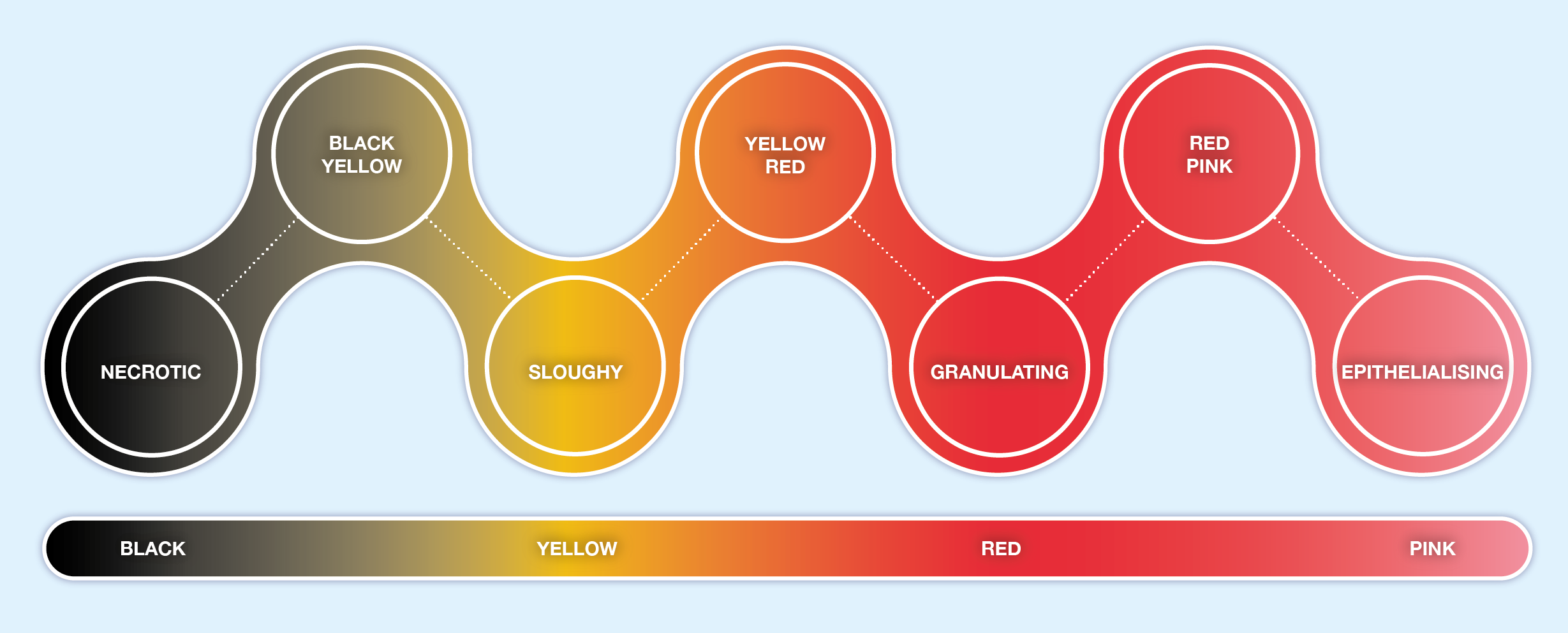
Figure 3.8 Wound Healing Continuum (derived from Gray et al, 2005)
It must be noted that this approach has been criticised for being too simplistic as wound healing is a continuum and wounds often contain a mixture of tissue types. The wound healing continuum is an effective audit tool and is an aid to clinical judgement making, it does not replace sound clinical judgement (Gray et al, 2010). However, it is still useful to be able to identify the 4 main types of tissue found during wound healing, whilst recognising, and giving consideration to, the possibility that more than one tissue type will coexist in a wound as it heals.
To decide upon the correct care and management of a wound and appropriate dressing selection it is necessary to identify the tissue type. A wound may have a variety of these tissues present at any one time.
The tissue type within the wound will determine the primary aim of treatment (see Module 4). The percentage of tissue types present in a wound should be recorded during the initial assessment. Any changes in the percentages can act as a marker of wound improvement or deterioration (Eagle, 2009).
BLACK (NECROTIC TISSUE)
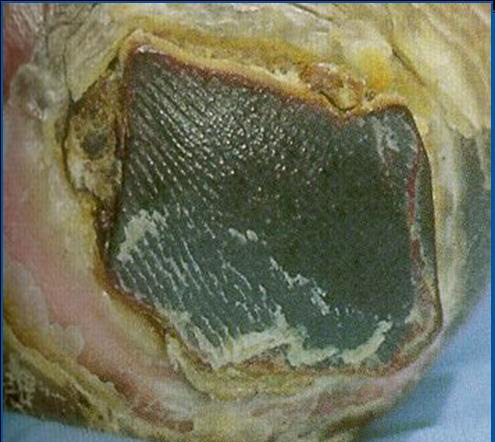
Fig 3.9 Patient showing signs of necrosis
NB: Necrotic tissue on the feet and leg lower limb, (including feet) should be treated with great caution. The wound should be covered with a dry dressing and a vascular status established before treatment is carried out. Referral to the relevant speciality e.g. vascular or diabetic foot clinic as per local policy. Delaying referral could threaten the limb (Eagle, 2009).
Management of necrosis should include identifying the underlying cause of the necrosis e.g. pyoderma gangrenosum, peripheral arterial disease, calciphylaxis, insect /snake bite, and implementing the appropriate treatment before the dead tissue can be dealt with. This can encompass administering antibiotics, anti-venom and or alleviating pressure on the wound area to restore perfusion. (Carpenter 2017, Wound Source 2019).
Once the cause has been established the necrotic tissue, where appropriate, will need to be removed so that wound healing can begin. Unless removed the eschar will delay healing as healing cannot proceed effectively without a moist wound environment. The necrotic tissue is derived from granulation tissue after the death of fibroblasts and endothelial cells (Collier, 2004).
Caution should be applied with wounds showing signs of pyoderma gangrenosum and calciphylaxis, a referral to a specialist healthcare professional for further advice may be required (Carpenter 2017).
Necrotic tissue also acts as a culture, providing an ideal breeding ground for bacteria (Eagle, 2009). Removing this tissue will also allow for accurate assessment of the wound bed as the necrotic eschar can mask the true size of the wound.
The presence of necrotic tissue presents a challenge for healthcare practitioners. The removal of necrotic devitalised tissue can be achieved by a variety of evidenced-based interventions. See module 4. Removal can be achieved with dressings that rehydrate the hard tissue. Larvae can be used if the necrotic tissue is soft. Within the nursing profession conservative sharp debridement, defined as the removal of dead tissue with a scalpel above the level of viable tissue has been identified as a high risk clinical procedure.
YELLOW (SLOUGH)
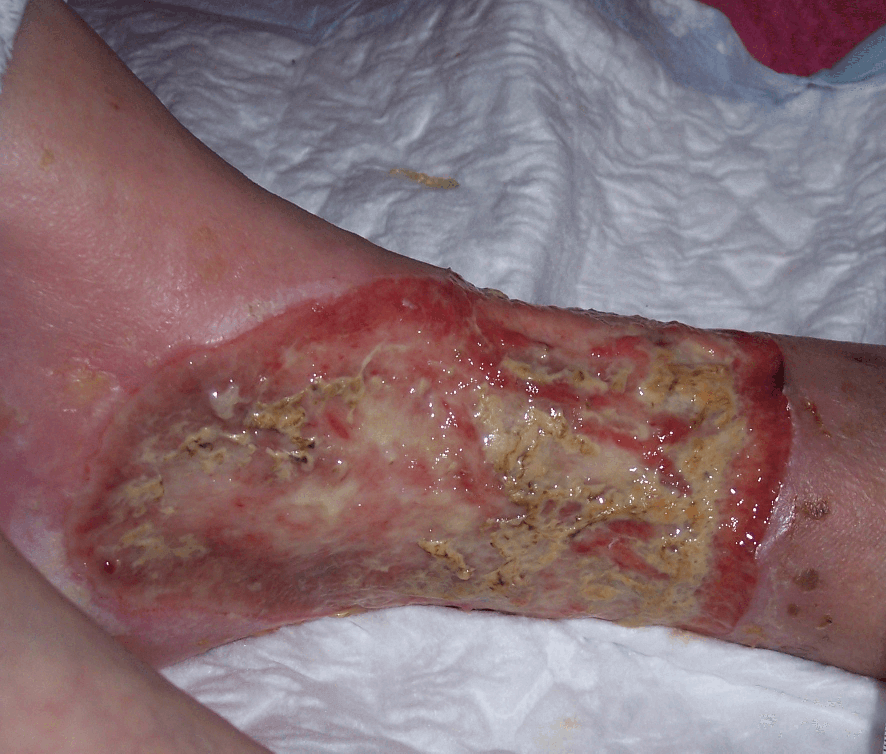
Sloughy tissue (Figure 3.10) is fibrous and yellow, adheres to the wound bed and cannot be removed on irrigation (Collier, 2004). It is also a type of necrotic tissue. Slough consists of dead cells and wound debris which should be removed to enable healing to take place. This is often referred to as ‘de-sloughing’.
Slough may predispose to wound infection and delay healing, however the presence of slough is not necessarily indicative of clinical infection. Exposed tendons should not be mistaken for slough.
Slough can be found as patches across the wound bed. Removal of the slough is done so by the application of a suitable dressing which enables the wound to progress to healing (Eagle, 2009).
RED (GRANULATION)
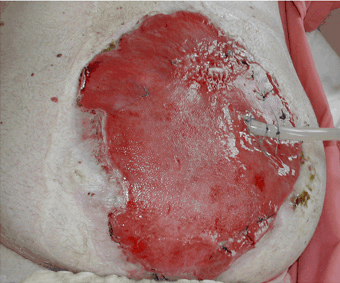
Fig 3.11 An example of a granulating wound
The aim of wound management for a granulating wound is to;
- Manage excess wound exudate
- Protect from infection
- Maintain a moist wound environment for wound healing
- Encourage growth of new tissue (Eagle, 2009)
PINK (EPITHELIALISATION)
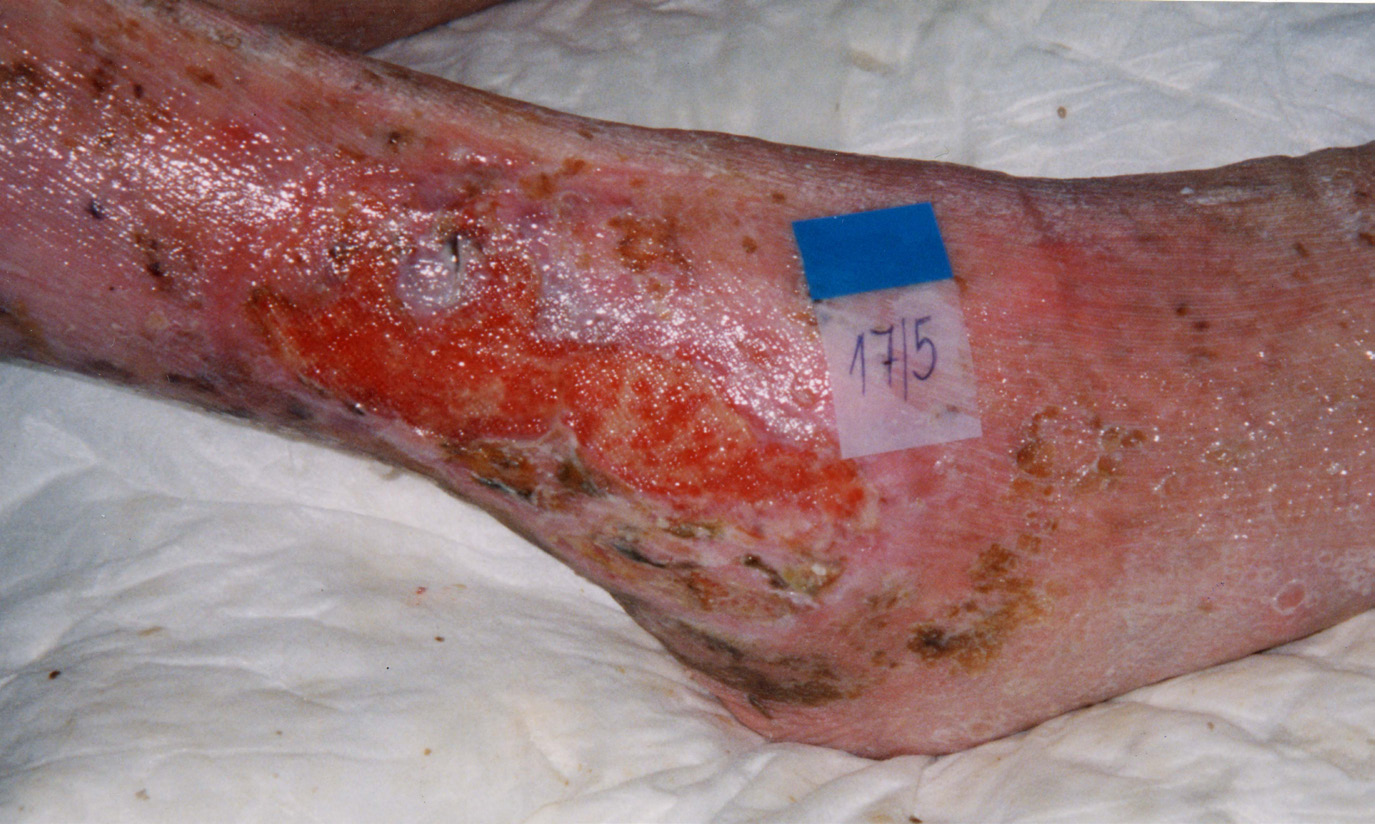
In shallow wounds with a large surface area, islets of epithelialisation may be seen.
INFECTION
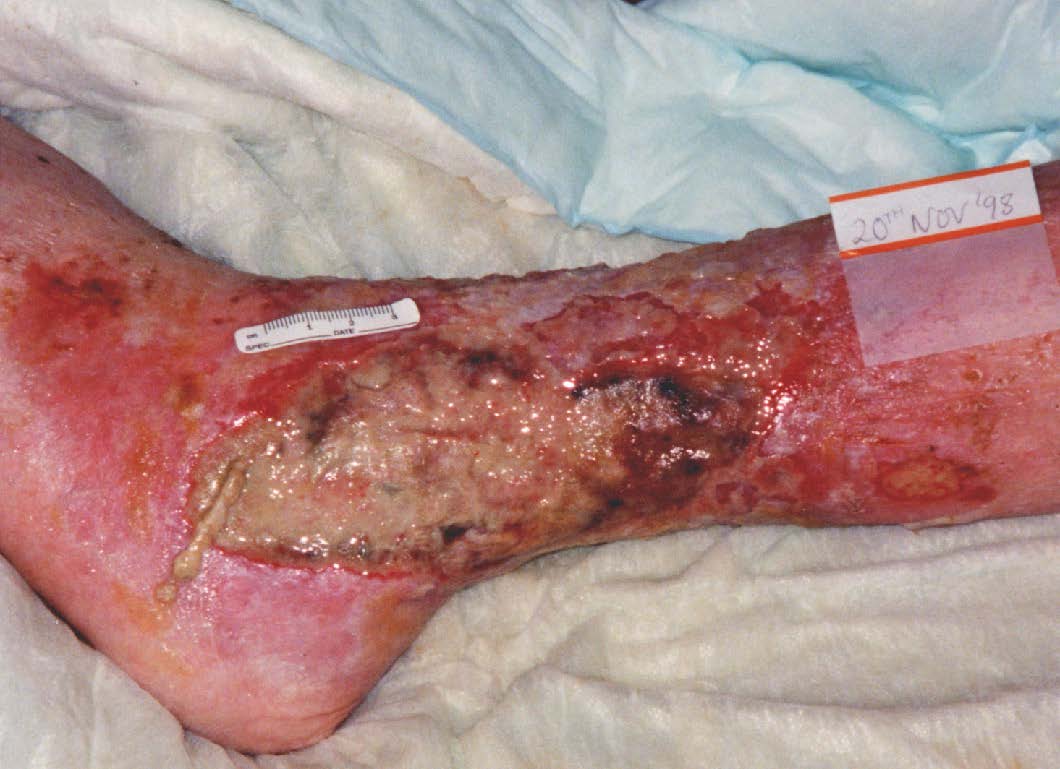
Fig 3.13 An example of an infected wound
Wound infection and associated delayed healing present considerable challenges for clinicians, particularly with respect to identifying clinical infection and choosing appropriate treatment options (EWMA, 2005). Clinical signs of infection include erythema, swelling, local warmth/heat and pain. (EWMA, 2005).
For further information on tissue types see Module Four, Section 3
Criteria for Identifying Wound Infection
Criteria:
- Abscess
- Cellulitis
- Discharge (serous exudate with inflammation; seropurulent; haemopurulent; pus)
Suggested Additional Criteria:
- Delayed healing (compared with normal rate for site/condition)
- Discolouration of friable granulation tissue that bleeds easily
- Unexpected pain/tenderness
- Pocketing at base of wound
- Bridging of the epithelium or soft tissue
- Abnormal smell
- Wound breakdown
(Cutting & Harding, 1994; Dowsett 2011)
The normal microbial state of a healing wound is that of colonisation, which represents a stable state where growth and death of organisms is balanced or below the immune systems healing disruption threshold (Heinzelmann et al, 2002),
The management of infected wounds will be discussed further in Module Four, Section 4: Infection.
